CORONA STUDY USING DELTAREX-G FOR COVID-19
Available now: “Right to Try” DeltaRex-G for COVID-19 EMERGENCY USE
Coronavirus disease 2019 (COVID-19) is an infectious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). This aggressive and highly infectious virus can lead to life threatening complications including cytokine storm (raging fevers, headaches, intense fatigue, muscle aches, loss of taste and smell) and acute respiratory distress syndrome (difficulty breathing from pneumonia leading to respiratory failure). These complications are the main causes of death in this global pandemic. However, these complications are believed to be caused not by the virus itself, but by an exaggerated immune reaction to the infection. The overactive immune cells are thought to make certain proinflammatory chemicals (cytokines) that cause raging fevers and damage to the lungs and other organs.
Over 1000 clinical trials are on-going worldwide to diagnose, treat, and improve the rate of recovery from COVID-19. So far, there are a few promising drugs but no FDA-approved treatment for COVID-19. We announce DeltaRex-G, the first, and so far, only, gene therapy for treatment of COVID-19 induced Cytokine Storm and Respiratory Distress Syndrome.
WHY DELTAREX-G FOR COVID-19?
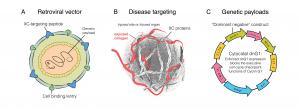
1) DeltaRex-G has a navigational system that seeks out areas of injury (e.g. inflamed lung), thus increasing the drug concentration in injured sites, where overactive immune cells are creating havoc. Our hypothesis is that DeltaRex-G can hasten recovery from pneumonia/ARDS and prevent chronic lung disease by killing the activated immune cells causing ARDS and the proliferative fibroblasts causing chronic lung disease;
2) DeltaRex-G has been given to over 280 cancer patients with few, if any, toxic side effects; and
3) DeltaRex-G is already FDA approved for the Foundation’s “Right-to-Try” Expanded Access Program as treatment of Stage 4 cancer.
WHAT IS THE STUDY ABOUT?
The CORONA STUDY is a Phase 1/2 study that will test whether DeltaRex-G is safe, will improve the symptoms of COVID-19, will hasten patient recovery from pneumonia/ARDS and will prevent chronic lung disease induced by COVID-19.
The Aveni Foundation (www.avenifoundation.org) is raising $5 Million USD for this upcoming study: The CORONA study Using DeltaRex-G for COVID-19 (www.clinicaltrials.gov: NCT04378244). However, available now is a limited supply of DeltaRex-G for “Right to Try” EMERGENCY USE.
TO DONATE: go to www.avenifoundation.org, click on CONTRIBUTE, or write a check payable to: Aveni Foundation, 2811 Wilshire Blvd., Suite 777, Santa Monica CA 90403; or Wire funds to: Chase Bank, Account Name: Aveni Foundation, Routing No: 322 271 627, Checking Acct. No.: 317 312 673, SWIFT CHASUS33.
For more information, email Dr. Gordon at egordon@avenifoundation.org or egordon@sarcomaoncology.com.
Erlinda Maria Gordon
Aveni Foundation
+1 818-726-3278
email us here
Erlinda M. Gordon, MD- DeltaRex-G Targeted Gene Therapy for COVID-19
EIN Presswire does not exercise editorial control over third-party content provided, uploaded, published, or distributed by users of EIN Presswire. We are a distributor, not a publisher, of 3rd party content. Such content may contain the views, opinions, statements, offers, and other material of the respective users, suppliers, participants, or authors.